Two-thirds of stroke survivors never fully regain hand function. Not because the intent to move disappears — in many patients, the brain still fires the signal — but because the pathway between intent and muscle has been severed or damaged. For decades, rehabilitation has meant repetition therapy, hoping the brain rewires itself fast enough before the recovery window closes around the six-month mark. Michel Maharbiz, Ph.D., founder of iota Biosciences and former UC Berkeley professor, thinks the window can be forced open with the right hardware.
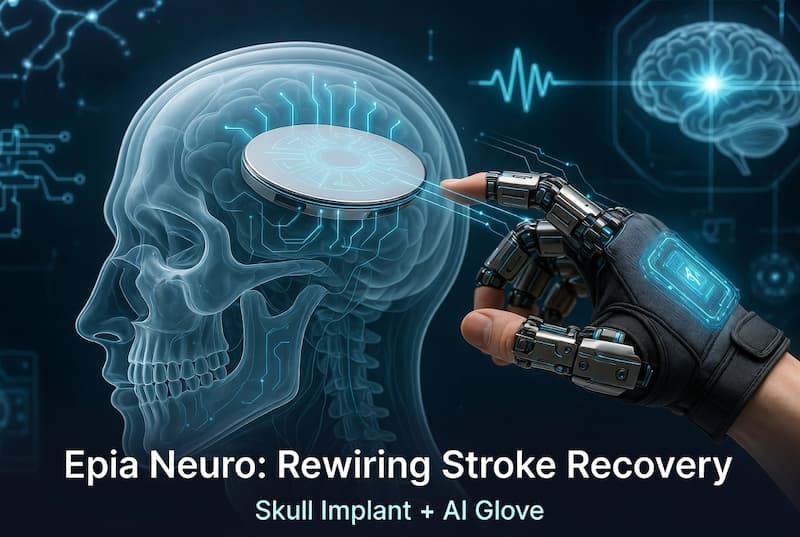
On April 2, 2026, his company Epia Neuro emerged from stealth with a system that pairs a disk-shaped skull implant with an AI-guided motorized grip glove. The goal isn’t to build a permanent prosthetic bypass. It’s to use the device itself to drive neuroplasticity — strengthening the brain’s own motor pathways until the patient no longer needs the glove at all.
That’s a fundamentally different bet from the rest of the BCI field, and it might be the right one.
The Grip Problem Is the Bottleneck for Stroke Independence
Stroke survivors frequently describe the same frustration: cognition intact, motivation intact, but the hand won’t cooperate. Buttoning a shirt, holding a fork, opening a door — all gated behind a grip that no longer reliably responds. Approximately two-thirds of stroke survivors experience persistent hand and arm impairment significant enough to affect daily independence.
Standard therapies plateau. After roughly six months post-stroke, conventional rehabilitation — physical therapy, occupational therapy, constraint-induced movement therapy — tends to deliver diminishing returns. The neural damage doesn’t reverse; at best, the brain slowly finds alternate routes, a process called cortical remapping. Epia Neuro’s premise is that you can accelerate and strengthen this remapping by giving the brain immediate, high-fidelity feedback every time it attempts to move.
The concept is called closed-loop neural rehabilitation: detect the intent signal, convert it into real movement via the glove, and let the brain register the consequence. Repeat thousands of times. The Hebbian principle — neurons that fire together wire together — does the rest.
A Skull Implant That General Neurosurgeons Can Place in Under an Hour
The implant itself is designed to sit inside the skull, replacing a small bone flap, without penetrating the dura — the tough protective membrane that wraps the brain. This matters enormously for risk profile. Penetrating electrodes (as used in Neuralink or Blackrock Utah arrays) offer higher bandwidth but introduce scarring risk, signal degradation over time, and demand highly specialized surgical teams.
Epia’s approach trades some raw signal resolution for a dramatically lower procedural burden. The implantation is designed to take under one hour and to be performable by general neurosurgeons in standard hospital settings — not just elite neurosurgery centers in Boston or San Francisco. No chest implant. No tunneled leads running under the skin. The implant recharges non-invasively via a headset every few days.
Critically, the skull cradle is built for upgradability: future hardware improvements can replace the internal module without additional invasive surgery. That’s a long-term platform bet, not a one-shot device. The company has already signaled future indications beyond stroke — Parkinson’s disease, epilepsy, and cognitive decline — all leveraging the same read/write neural architecture.
The AI Layer: Learning to Decode Intent from Noisy Signals
The implant captures neural signals. But raw intracranial signals are notoriously difficult to interpret — variable between patients, shifting over time, influenced by fatigue, attention, and medication. This is where the system’s AI layer does the real work.
Sensors embedded in the motorized glove capture physical context — hand position, resistance, attempted movement — and feed that data back to the decoding algorithm alongside the neural signal. The system learns each patient’s unique neural signature for grip intent, continuously recalibrating as the brain itself changes during rehabilitation. In machine learning terms, this is a personalized, adaptive decoder trained on a data distribution that shifts as the therapy succeeds.
Chief Technology and Science Officer Gil Mandelbaum, Ph.D. leads the neuroscience and engineering integration. The technical foundation builds directly on Maharbiz’s prior work at iota Biosciences, which was acquired by Astellas — a lineage that provides both credibility and IP continuity.
Where Epia Sits in the BCI Competitive Map
The BCI field in 2026 has three rough tiers defined by invasiveness and capability:
- Penetrating arrays (Neuralink, Blackrock): Highest bandwidth, deepest brain access, highest surgical complexity and risk. Designed for paralysis, ALS, and high-bandwidth communication tasks.
- Vascular approaches (Synchron Stentrode): Threaded through blood vessels into motor cortex, avoiding open-skull surgery. Lower signal quality, but extremely low procedural risk.
- Non-invasive or semi-invasive (EEG headsets, IpsiHand glove): No implant at all, or surface-only. Easiest to deploy, but signal quality degrades significantly through skull and scalp tissue.
Epia carves a specific niche: better signal quality than non-invasive approaches, substantially lower surgical burden than penetrating arrays, and a rehabilitation-first philosophy that distinguishes it from systems optimized for permanent substitution. Neurologist advisor David J. Lin frames the distinction clearly — using the device drives plasticity, so native arm and hand function can improve even after the glove is removed. The device is meant to make itself unnecessary.
2026 Trials and the Road to Real-World Deployment
Epia Neuro’s first-in-human demonstration is planned for later in 2026 at Lenox Hill Hospital in New York, with additional trial sites coming online by year-end. The company is led by Maharbiz as CEO, Mandelbaum as CTSO, and Michelle Patruno as COO — a team with complementary backgrounds in neuroscience, hardware, and operational scaling.
The trial results will answer the critical question the field has circled for years: can a minimally invasive intracranial device, deployed outside specialized centers, produce measurable, durable functional gains in a large stroke population? If yes, the implications extend well beyond grip. A platform that general neurosurgeons can implant in under an hour, that upgrades without reopening the skull, and that routes through AI-adaptive decoding is infrastructure for the next decade of neurorehabilitation — not just a single-indication device.
Neuroplasticity Is the Feature, Not the Side Effect
Most BCIs are designed around substitution: the brain can’t move the arm, so we reroute the signal to a robotic limb or a cursor. Epia Neuro is designed around restoration: the brain can still generate the signal, so we amplify it into real movement until the brain learns to do it alone.
That’s a harder engineering problem, a harder clinical trial problem, and a harder commercial story. But if the 2026 human trials bear out the neuroplasticity hypothesis — if patients who complete rehabilitation with the Epia system show measurable improvements in native hand function after removing the glove — the company will have proved something important. Not just that BCIs can substitute for lost function, but that they can genuinely return it.
That would change what we think BCIs are for.
References
- Epia Neuro. (2026). Official launch press release. BusinessWire. https://www.businesswire.com
- Mullin, E. (2026). A New Implant Aims to Rewire the Brain to Help Stroke Patients. WIRED. https://www.wired.com
- Epia Neuro. (2026). Company overview and platform description. epianeuro.com. https://www.epianeuro.com
Related: Neuralink’s VOICE Trial: Decoding Speech from Neural Signal · What Is a Biocomputer in 2026? · Biohybrid Robots: When Machines Grow Their Own Muscles
Feature image: AI-generated using Grok